|
An element is a pure substance made up of only one type of atom (the simplest. Each row is called a period, and each column is called a group. Sodium is a white-silvery metal that belongs to Group 1 (alkali metals) and Period 3 elements in the periodic table. Trends like this one demonstrate that electrons within atoms are organized in groups. The periodic table is a chart with all the known chemical elements. A high electronegativity value means an atom readily attracts electrons to form a chemical bond with another atom. Electronegativity is a measure of how easily an atom attracts a pair of electrons to form a chemical bond. This quantum-mechanical phenomenon determines the period lengths of 2, 8, 8, 18, 18, 32. Thus, it takes much more energy than just overcoming a larger ionic charge would suggest. Periodic table showing the trend from low to high of the element’s electronegativity. An important condition for the emergence of periodicity of chemical behavior under ambient conditions is a well-structured atomic orbital level scheme, in particular with gaps, above 1s and 2p to 6p. In the context of chemistry and the periodic table, periodicity refers to trends or recurring variations in element properties with increasing atomic number. Why is it so much larger? Because the first two electrons are removed from the 3 s subshell, but the third electron has to be removed from the n = 2 shell (specifically, the 2 p subshell, which is lower in energy than the n = 3 shell). Each next element in a period has one more proton and is less metallic than its predecessor. All elements in a row have the same number of electron shells. A period on the periodic table is a row of chemical elements. The rows are arranged so that metals are on the left side of the table and nonmetals are on the right side. In the periodic table of the elements, each numbered row is a period. 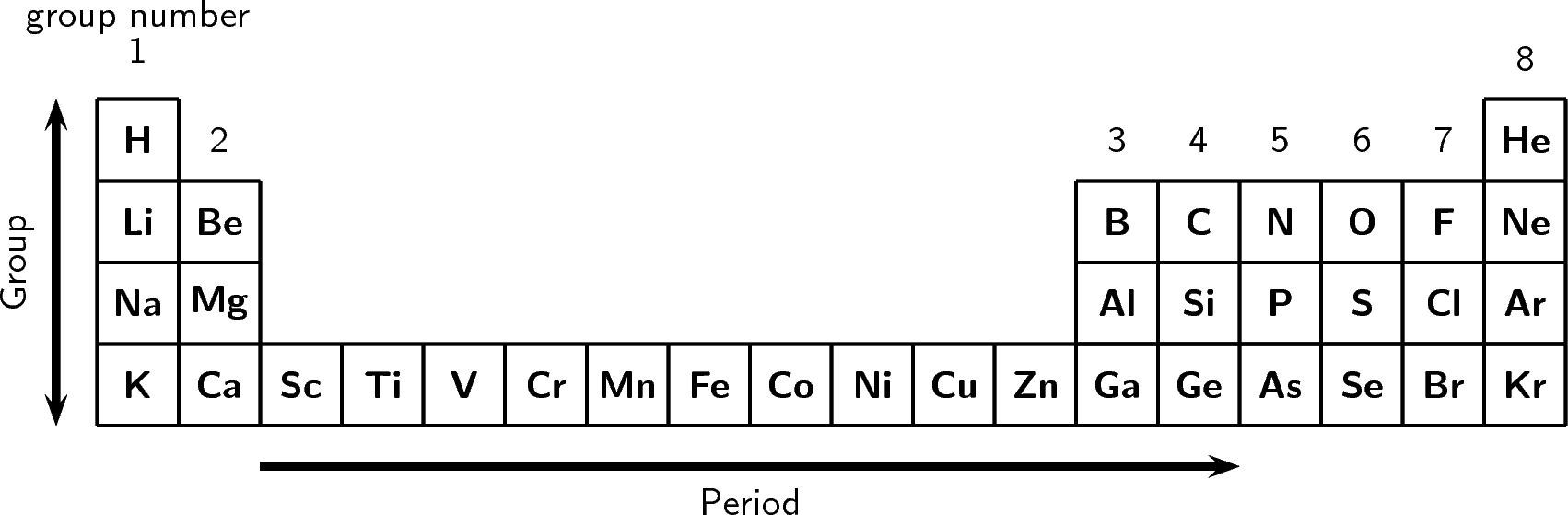
The seven rows of the periodic table are called periods. The third IE, however, is over five times the previous one. The periodic table is a tabular arrangement of chemical elements that is arranged by increasing atomic number and groups elements according to recurring properties. 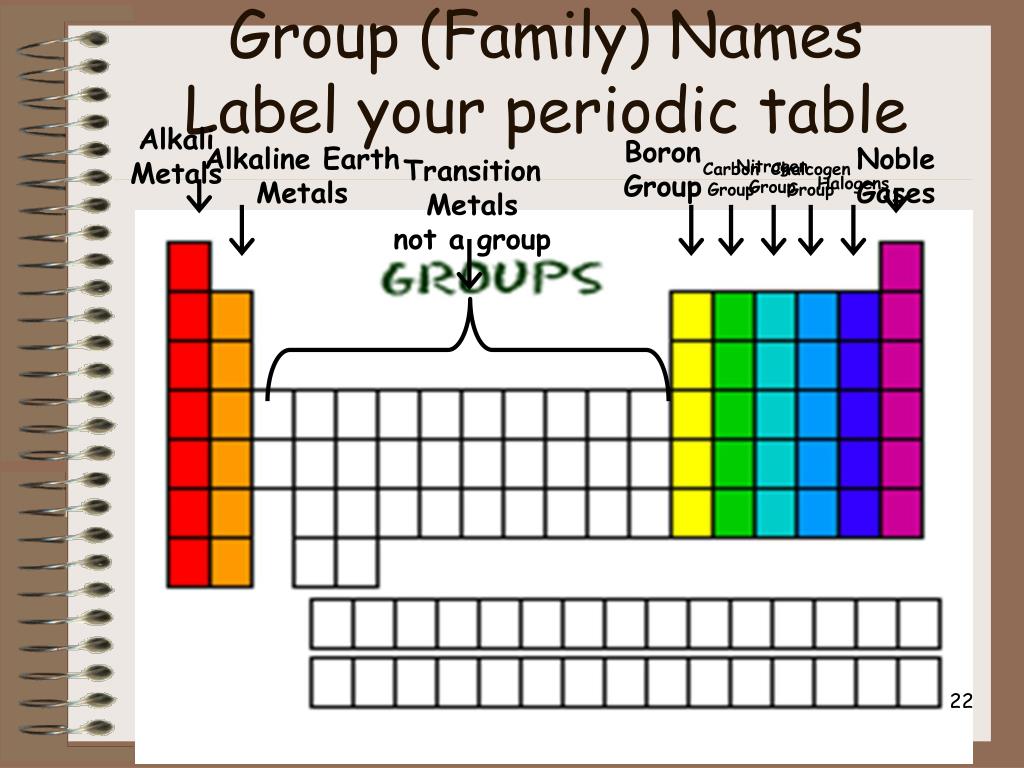 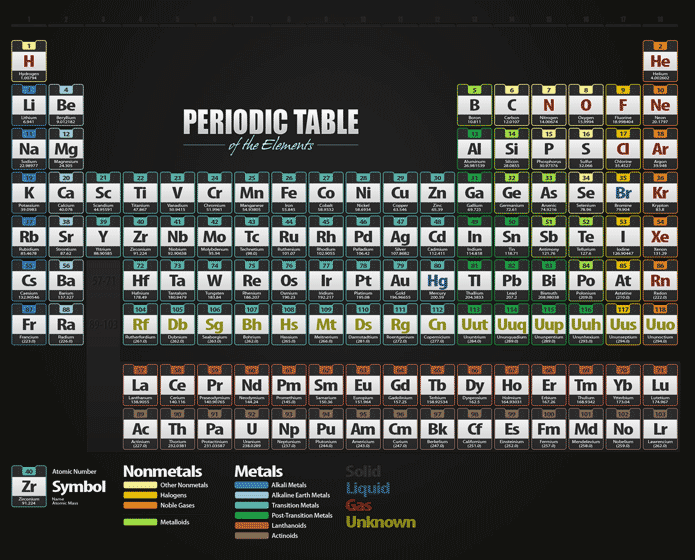
These changes occur within their respective period from left to right and group from top to bottom in the periodic. The alkali metals are so called because reaction with water forms alkalies (i.e., strong bases capable of neutralizing acids ). These observed patterns allude to the changes in atomic structure, including size and radius, as well as properties of the elements. alkali metal, any of the six chemical elements that make up Group 1 (Ia) of the periodic table namely, lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. Periodic trends are specific patterns observed among the chemical elements of the periodic table.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
